 
Thermal properties of water at different temperatures like density, freezing temperature, boiling temperature, latent heat of melting, latent heat of evaporation, critical temperature and more. TemperatureĬavitation of impellers increases with water temperatures.īoiling temperatures for common liquids and gases - acetone, butane, propane and more. Hot and cold water service systems - design properties, capacities, sizing and more.įeeding Pumps in Steam Systems - Suction Lift Head vs. Thermodynamics of steam and condensate systems. Steam & condensate systems- properties, capacities, pipe sizing, systems configuration and more. PSI, 1.01325 0.10132 14.696 CFM (cubic feet / minute) CFM CFM, l/min l/s m3/h, 28.32 0.472 1.699 ft3 (cubic foot) ft ft. Material properties of gases, fluids and solids - densities, specific heats, viscosities and more. 40 psi to atm 2.72184 bmw blue gloss wrap 2 Atm To Psi - M圜alcu. See also Water Density, specific weight and thermal expansion coefficient, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of Vaporization, Ionization Constant, pK w, of normal and heavy water, Pressure and Boiling Points, Specific gravity, Specific heat (heat capacity) and Specific volume for online calculatores, figures and tables.īoiling points of elements, products and chemical species at varying conditions. 2atm to psi Veeb1006.2 Atm to Psi conversion calculator converts 1006.2 atm into. 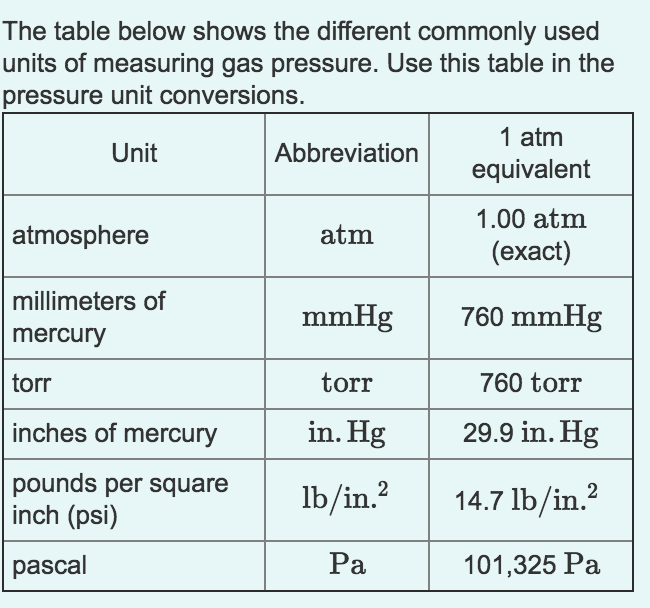
See Water and Heavy Water - for thermodynamic properties. See also other properties of Water at varying temperature and pressure: Boiling points at vacuum pressure, Density and specific weight, Dynamic and kinematic viscosity, Enthalpy and entropy, Heat of vaporization, Ionization Constant, pK w, of normal and heavy water, Melting points at high pressure, Prandtl number, Properties at Gas-Liquid Equilibrium Conditions, Saturation pressure, Specific gravity, Specific heat (heat capacity), Specific volume, Thermal conductivity, Thermal diffusivity and Vapour pressure at gas-liquid equilibrium. See Water and Heavy Water for thermodynamic properties at standard condtions. Note! Pressure must be within the ranges 1-220 bara, 14.7-3200 psia, 760-165 000 mm Hg or 30-6500 in Hg.īoiling points of water at absolute pressures ranging from 1 to 70 bara or 14.7 to 1000 psia are indicated in the figures and tables below: The output temperature is given as ☌, ☏, K and °R. The calculator below can be used to calculate the water boiling point at given absolute pressures. The formulas to describe the relationship are: PSIG + 1 atm PSIA and. Note that PSIG is always lower than PSIA. The boiling point of water depends on pressure. 1 psi 0.0689475729 bar 1 bar 14.503773773 psi Example: convert 15 psi to bar: 15 psi 15 × 0.0689475729 bar 1. Examples of How to Calculate PSIG and PSIA. When water is heated it reach a temperature - the boiling point - at which the vapor pressure is large enough that bubbles are formed inside the water.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
